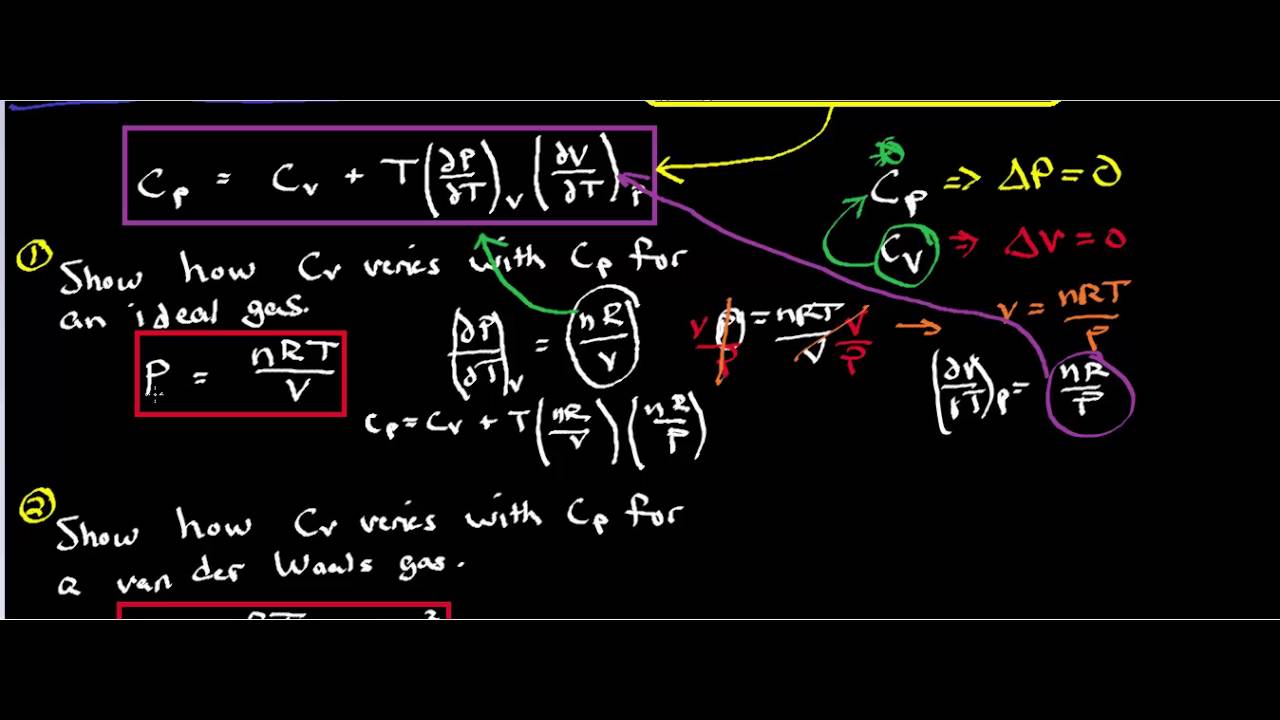
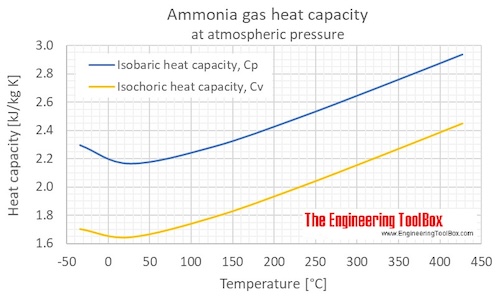
If CP and CV denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, then.

Cp/Cv=gamma Proof | Relation Between Specific Heats & Adiabatic Index | Properties of Gases | BME - YouTube

Development in the Concept of Bacterial Polysaccharide Repeating Unit-Based Antibacterial Conjugate Vaccines | ACS Infectious Diseases

Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S